Modified: July 31, 2021
cellular respiration
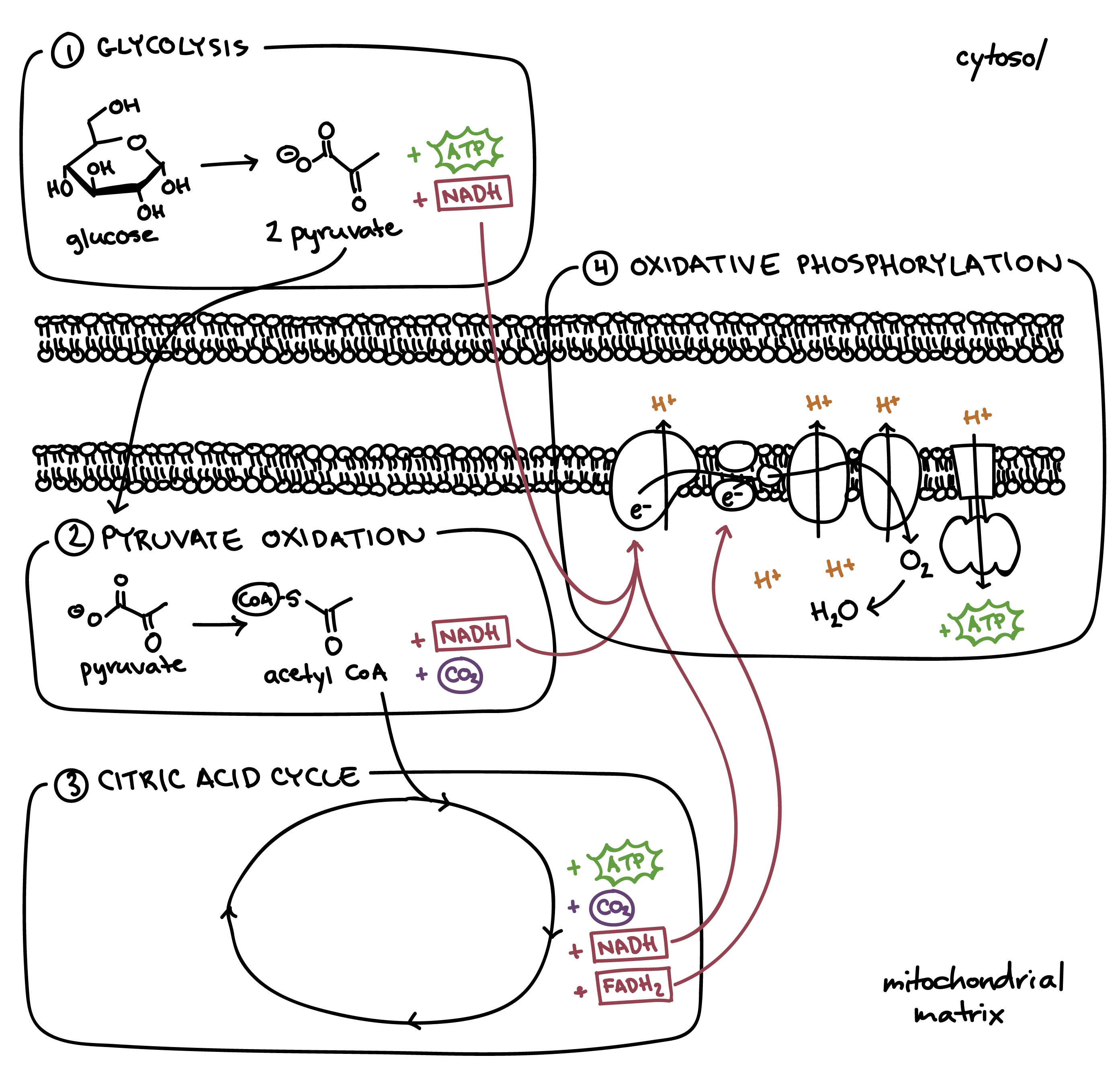
This page is from my personal notes, and has not been specifically reviewed for public consumption. It might be incomplete, wrong, outdated, or stupid. Caveat lector.We can view cellular respiration (and combustion more generally) from a high level as the transfer of electrons from carbon to oxygen atoms; in other words, oxidation of carbon atoms. We start with some hydrocarbon-ish thing in which a lot of carbons are bound to hydrogens. Maybe this is glucose, which also has some oxygen atoms bound up, but that doesn't change much. We also have some free O2 molecules. We end with water--in which oxygen takes the hydrogens---plus CO2, in which the oxygen atoms get to steal electrons from the carbon. Since oxygen really likes electrons, this is a lower-energy state than we started in, meaning that energy is released during the reaction.